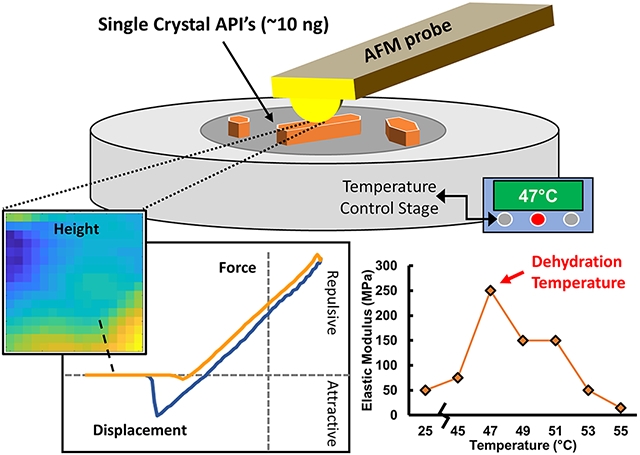
Atomic Force Microscopy to Identify Dehydration Temperatures for Small Volumes of Active Pharmaceutical Ingredients.
The environmental circumstances related to altering the hydration state of lively pharmaceutical components (API) are essential to understanding their stability, bioperformance, and manufacturability. Identifying the dehydration occasion utilizing < 1μg of materials is an more and more vital problem.
Atomic Force Microscopy indentation mapping is carried out at managed temperatures between 25-100°C, for nanoscale volumes of hydrated APIs exhibiting distinct dehydration conduct and anhydrous APIs as controls.
For caffeine hydrate and azithromycin dihydrate, the relative mechanical modulus will increase ~10-fold at dehydration temperatures.
These are confirmed by typical macroscopic measurements together with Variable Temperature Powder X-ray Diffraction, Thermogravimetric Analysis, and Differential Scanning Calorimetry.
Conversely, no such mechanical transition is noticed for anhydrous ibuprofen or a proprietary anhydrous compound. AFM-based mechanical mapping is due to this fact demonstrated for small-volume willpower of temperature-induced solid-state dehydration occasions, which can allow spatially or temporally mapping for future research of dehydration mechanisms and kinetics, as a operate of commercially related nanoscale heterogeneities.
Public reporting on pharmaceutical industry-led entry packages: alignment with the WHO drugs packages analysis guidelines.
There has been elevated demand for larger public accountability and transparency of non-public sector-led world well being partnership packages.
This examine critically evaluations and pilot checks the World Health Organization (WHO) drugs program guidelines as a framework for public reporting and assessing of packages.We reviewed every query on the WHO guidelines for readability and value.
Next, we pilot examined the subset of guidelines questions centered on program evaluation. We extracted and analyzed publicly obtainable data on one randomly chosen program from every of the 20 largest research-based biopharmaceutical corporations. For every program, we assessed whether or not publicly obtainable data allowed for an evaluation of every related query within the guidelines.
Checklist questions slot in 4 fundamental classes: [1] nationwide well being and growth plans, wants, capability, legal guidelines and insurance policies; [2] monetary, efficiency, and public accountability; [3] threat administration and mitigation methods; and [4] long-term sustainability. Nearly all (21 of 22) questions within the guidelines require data finest supplied by corporations; one query requires data finest supplied by governments.Programs incessantly reported on the general public well being wants of their packages (100%), program goals and actions (100%) and the precise or anticipated program outputs (95%).
There was much less data on program alignment with nation plans and capability (50%), detailed program monitoring and analysis plan (20%), dangers mitigation methods (5%), program wants evaluation (5%), and extra assets required from or contributed by authorities (0%).
The WHO guidelines of key concerns for evaluating proposals for entry to drugs packages could possibly be a helpful framework for public reporting of program data as most of guidelines questions ask for knowledge that ought to be obtainable to these main this system. Further revisions of the WHO guidelines will assist refine it to enhance readability and content material validity.