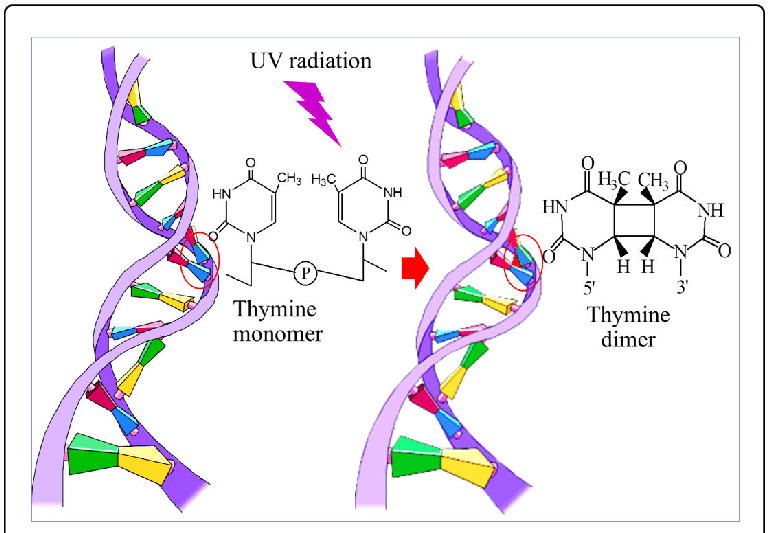
Thymine Dimer
Genprice Thymine Dimer products are available at Gentaur Europe from different suppliers at the best prices. Our products are of very high quality, validated by competent laboratories for exclusive use in research. You can order online or send your order to your local contact. If you are not sure of the product choice required for your research, you can always request a selection audit.
Thymine dimers
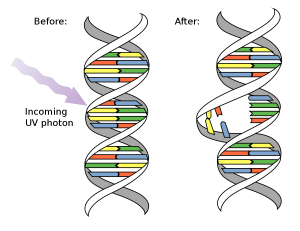
Summer is here and we all go outside to enjoy the sun. But remember to bring your sunscreen, as too much sunlight can damage your cells. Small doses of sunlight are needed to create vitamin D, but larger doses attack your DNA. UV light is the main culprit. The most energetic and dangerous wavelengths of ultraviolet light, called UVC, are scavenged (at least for now) by ozone in the upper atmosphere. However, weaker ultraviolet light, called UVA and UVB, passes through the atmosphere and is powerful enough to cause chemical changes in DNA.
Dangerous dimers
UV light is absorbed by a double bond in the thymine and cytosine bases in DNA. This added energy opens the bond and allows it to react with a neighbouring base. If the neighbour is another thymine or cytosine base, it can form a covalent bond between the two bases. The most common reaction is shown here: two thymine bases have formed a compact thymine dimer, with two bonds joining the bases. The top image is from the PDB entry and the bottom foreground image is from the PDB entry. This is not a rare event: every second you are in the sun, 50 to 100 of these dimers are formed in each skin cell!
Polymerase problems
These dimers are awkward and form a rigid kink in the DNA. This causes problems when the cell needs to replicate its DNA. DNA polymerase has trouble reading the dimer as it does not easily fit into the active site. TT dimers like the ones shown here are not the main problem, as they usually pair correctly with adenine when DNA is replicated. But CC dimers don’t fare as well. DNA polymerase often mispairs adenine with them instead of guanine, causing a mutation. If this occurs in an important gene that controls cell growth, such as the genes for tyrosine kinase Src or the p53 tumour suppressor, the mutation can lead to cancer.
Error handling
We spend a lot of time in the sun, so it’s no surprise that we have a powerful mechanism to correct these problems. Our cells use a process called nucleotide excision repair, which requires the concerted effort of a large collection of proteins that recognize the corrupt bases, cut out the section of DNA with the error, and then build a new copy of the damaged area. Other organisms have additional correction mechanisms. For example, the enzyme is an endonuclease that cleaves off damaged bases, making the site available for repair. Surprisingly, this endonuclease does not recognize the thymine dimer directly.
Instead, the enzyme recognizes one of the adenines that is paired with the dimer. Since the base pair is weakened by the contorted shape of the dimer, adenine easily flips over and binds to a pocket in the enzyme. The enzyme is a photolyase that directly breaks the bonds connecting the dimer, correcting the error in place. Ironically, photolyases use visible light to drive this process. This structure captures the DNA after the thymine dimer has been attached. Note that the two thymine bases (coloured in magenta) break off from the normal DNA helix and bind in a pocket on the enzyme surface.
Exploring the structure
Most DNA polymerases have difficulty replicating DNA with pyrimidine dimers. The enzyme on the left, from the entry of the PDB, is an exception: it is designed to read through damaged DNA. It has a loose active site, so it can easily accommodate the rigid thymine dimer. However, this open active site makes the enzyme quite error-prone. A more typical DNA polymerase is shown on the right, from the PDB entry. It tightly wraps around the DNA, and because of this close contact, it is very precise in copying the DNA. However, it has many problems with damaged bases and works 3000 times slower with thymine dimers than with normal DNA.