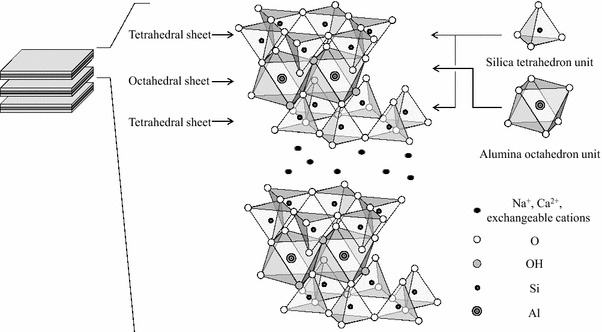
Application of montmorillonite in bentonite as a pharmaceutical excipient in drug delivery systems.
Montmorillonite is a multifunctional clay mineral and a main element of bentonite. Montmorillonite has been used in numerous industrial and pharmaceutical fields as a result of its distinctive traits, which embody swelling and adsorption.
The excessive adsorption capability of montmorillonite contributes to extend drug entrapment and sustained-release of medicine.
Montmorillonite typically sustains drug launch in many formulations by strongly adsorbing to the drug. In addition, montmorillonite enhances the dissolution fee and bioavailability of hydrophobic medicine.
Moreover, montmorillonite was utilized to kind composites with different polymer-based delivery techniques. Thus, montmorillonite might be utilized to formulate numerous drug delivery techniques to regulate and/or enhance the pharmaceutical properties of medicine, together with solubility, dissolution fee, and absorption. In this evaluation, views of making use of montmorillonite as a pharmaceutical excipient in drug delivery techniques are mentioned.
Study of In Vitro and In Vivo Carbamazepine Release from Coarse and Nanometric Pharmaceutical Emulsions Obtained through Ultra-High-Pressure Homogenization.
In the previous decade, pharmaceutical nanotechnology has confirmed to be a promising various for enhancing the physicochemical and biopharmaceutical options for standard pharmaceutical drug formulations. The purpose of this research was to develop, characterize, and consider the in vitro and in vivo launch of the mannequin drug carbamazepine (CBZ) from two emulsified formulations with totally different droplet sizes (coarse and nanometric). Briefly, oil-in-water emulsions have been developed utilizing
(i) Sacha inchi oil, ultrapure water, TweenTM 80, and SpanTM 80 as surfactants, (ii) methyl-paraben and propyl-paraben as preservatives, and
(iii) CBZ as a nonpolar mannequin drug. The coarse and nanometric emulsions have been ready by rotor-stator dispersion and ultra-high-pressure homogenization (UHPH), respectively. The in vitro drug launch research have been performed by dialysis, whereas the in vivo drug launch was evaluated in New Zealand breed rabbits.
The outcomes confirmed that nanoemulsions have been bodily extra steady than coarse emulsions, and that CBZ had a very low launch for in vitro willpower (<2%), and a launch of 20% in the in vivo research. However, it was discovered that nanoemulsions might considerably enhance drug absorption time from 12 h to 45 min.